
β-Diketimine appended periodic mesoporous organosilica as a scaffold for immobilization of palladium acetate: An efficient green catalyst for Wacker type reaction - ScienceDirect
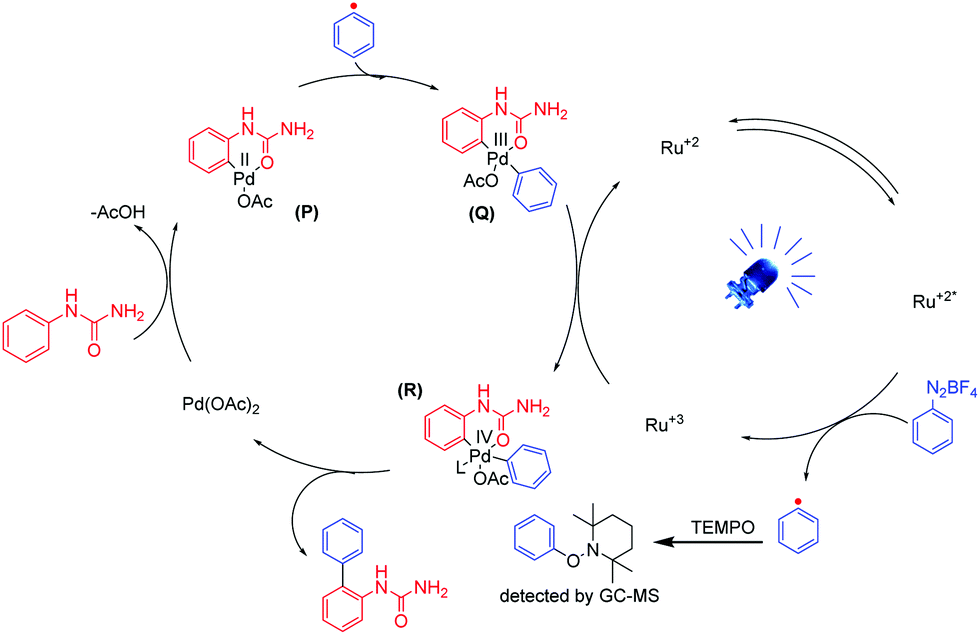
Dual palladium–photoredox catalyzed chemoselective C–H arylation of phenylureas - Chemical Communications (RSC Publishing)

Palladium in cephalosporin chemistry: an inexpensive triflate replacement for palladium acetate mediated coupling reactions - ScienceDirect
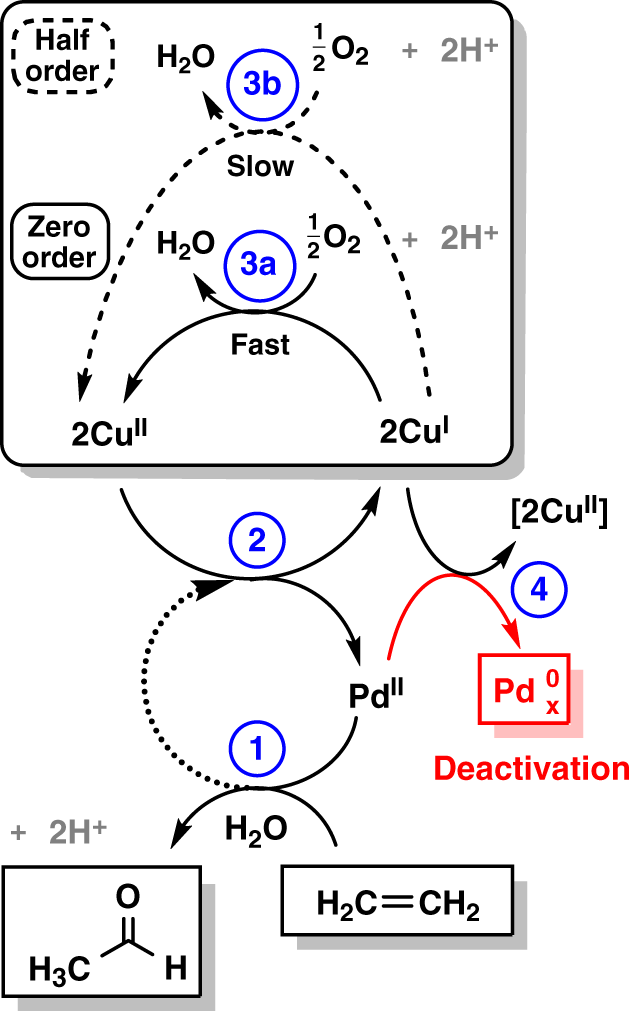
Elucidating the mechanism of heterogeneous Wacker oxidation over Pd-Cu/zeolite Y by transient XAS | Nature Communications
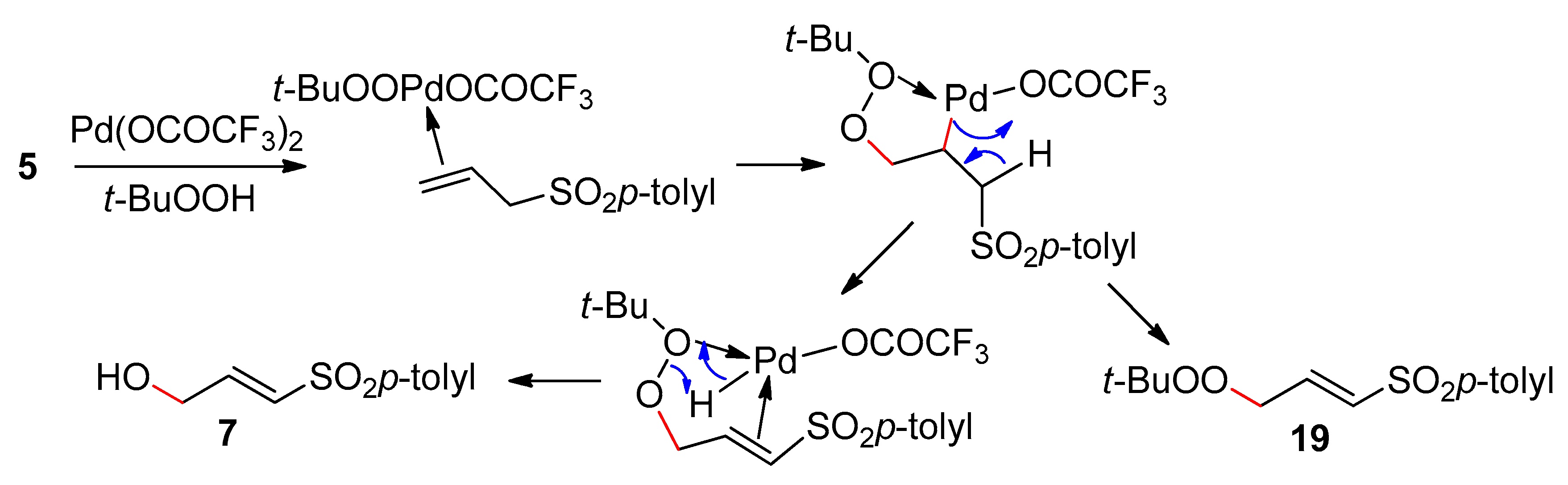
Catalysts | Free Full-Text | The Reims Journey Towards Discovery and Understanding of Pd-Catalyzed Oxidations | HTML
Coordination Chemistry Reviews Silica-supported palladium: Sustainable catalysts for cross-coupling reactions
MECHANISTIC STUDIES ON PALLADIUM-CATALYZED COUPLING REACTIONS BY BRADLEY PATRICK CARROW DISSERTATION Submitted in partial fulfil
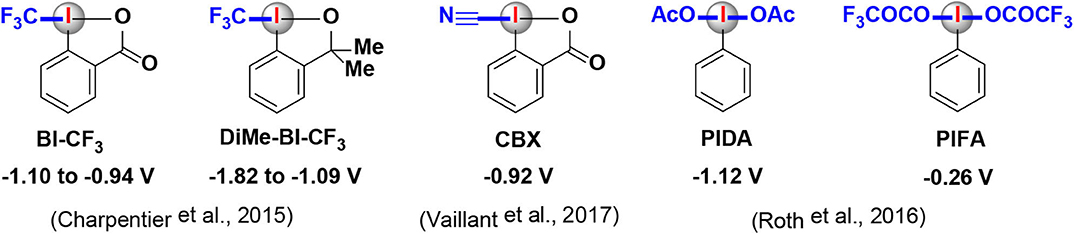
Frontiers | Recent Synthetic Applications of the Hypervalent Iodine(III) Reagents in Visible-Light-Induced Photoredox Catalysis | Chemistry
Coordination Chemistry Reviews Silica-supported palladium: Sustainable catalysts for cross-coupling reactions
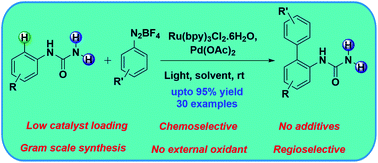
Dual palladium–photoredox catalyzed chemoselective C–H arylation of phenylureas - Chemical Communications (RSC Publishing)














