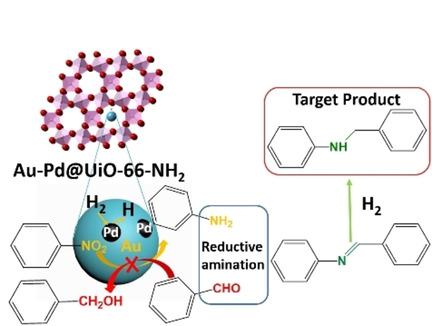
Pd‐Catalyzed Reductive Cyclization of Nitroarenes with CO2 as the CO Source - Guan - 2020 - European Journal of Organic Chemistry - Wiley Online Library
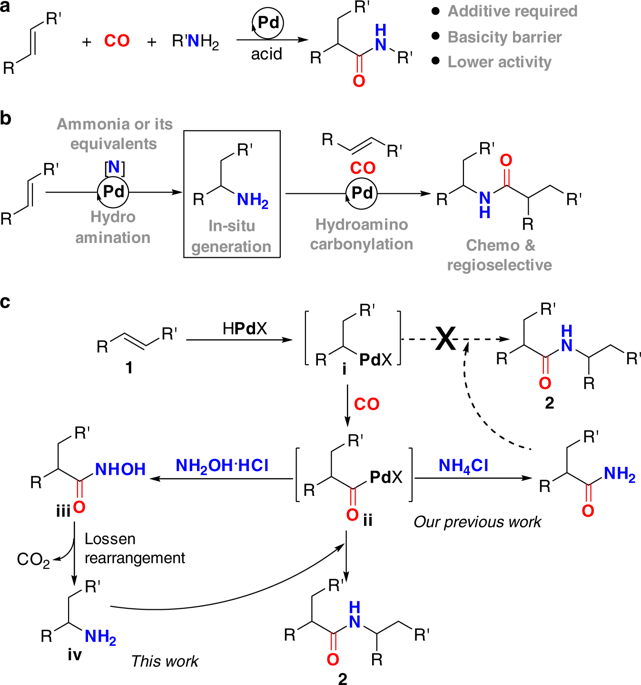
Palladium-catalyzed relay hydroaminocarbonylation of alkenes with hydroxylamine hydrochloride as an ammonia equivalent - Commun. Chem. - X-MOL

Table 1 from Convenient iron-catalyzed reductive aminations without hydrogen for selective synthesis of N-methylamines | Semantic Scholar

WO2016096905A1 - Synthesis of amides and amines from aldehydes or ketones by heterogeneous metal catalysis - Google Patents
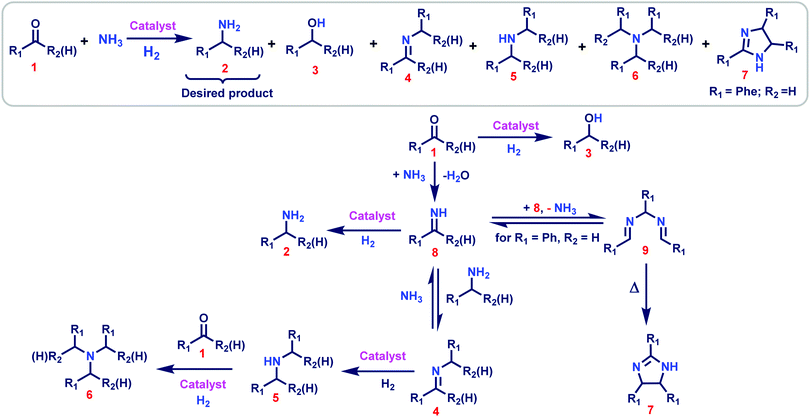
Catalytic reductive aminations using molecular hydrogen for synthesis of different kinds of amines - Chemical Society Reviews (RSC Publishing)

WO2016096905A1 - Synthesis of amides and amines from aldehydes or ketones by heterogeneous metal catalysis - Google Patents

Frontiers | Catalytic Reductive Amination of Aldehydes and Ketones With Nitro Compounds: New Light on an Old Reaction | Chemistry

WO2016096905A1 - Synthesis of amides and amines from aldehydes or ketones by heterogeneous metal catalysis - Google Patents
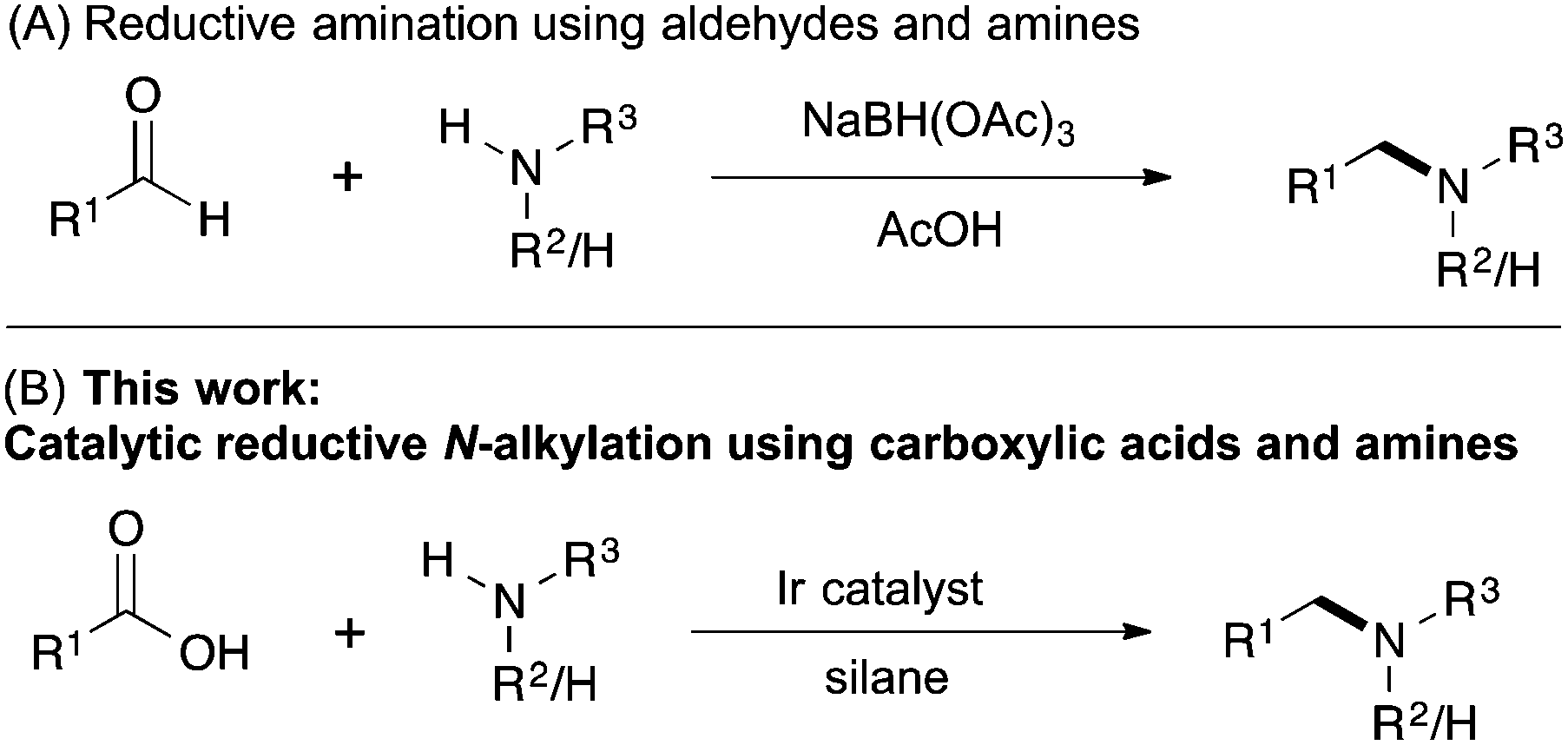
Catalytic reductive N-alkylation of amines using carboxylic acids - Chemical Communications (RSC Publishing)

AuPd−Fe3O4 Nanoparticle Catalysts for Highly Selective, One‐Pot Cascade Nitro‐Reduction and Reductive Amination - Cho - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

PDF) ChemInform Abstract: A Modified Palladium-Catalyzed Reductive Amination Procedure | Valerio Berdini, Alessandra Topai, and Giuseppe Nano - Academia.edu

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Simple ruthenium-catalyzed reductive amination enables the synthesis of a broad range of primary amines | Nature Communications