Stereocontrolled synthesis of enantiomerically pure 2-dienyl sulfoxides via palladium-catalyzed coupling reactions - ScienceDirect

PDF) First Synthesis of β-Keto Sulfoxides by a Palladium-Catalyzed Carbonylative Suzuki Reaction | Gregorio Asensio - Academia.edu
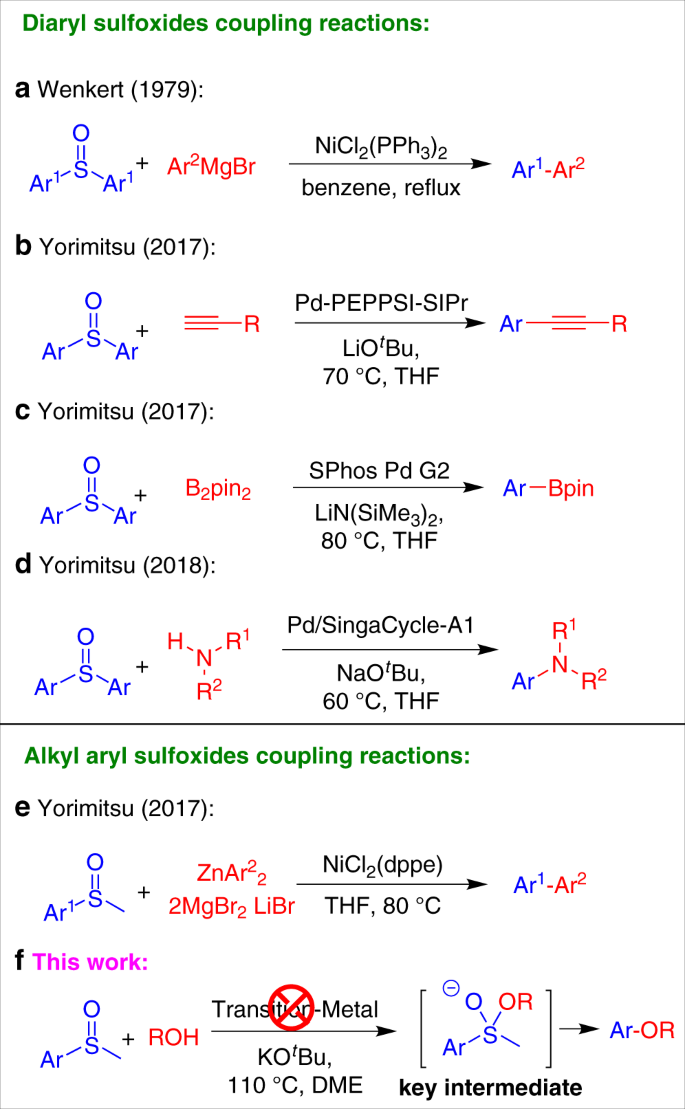
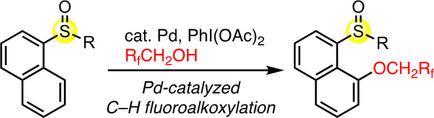
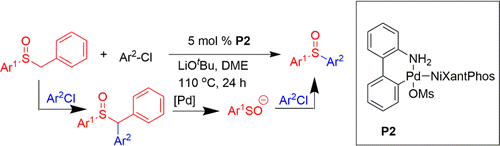
Transition-metal-free formal cross-coupling of aryl methyl sulfoxides and alcohols via nucleophilic activation of C-S bond | Nature Communications
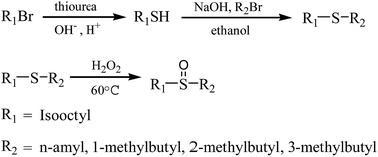
Solvent extraction of palladium(ii) with newly synthesized asymmetric branched alkyl sulfoxides from hydrochloric acid - RSC Advances (RSC Publishing)
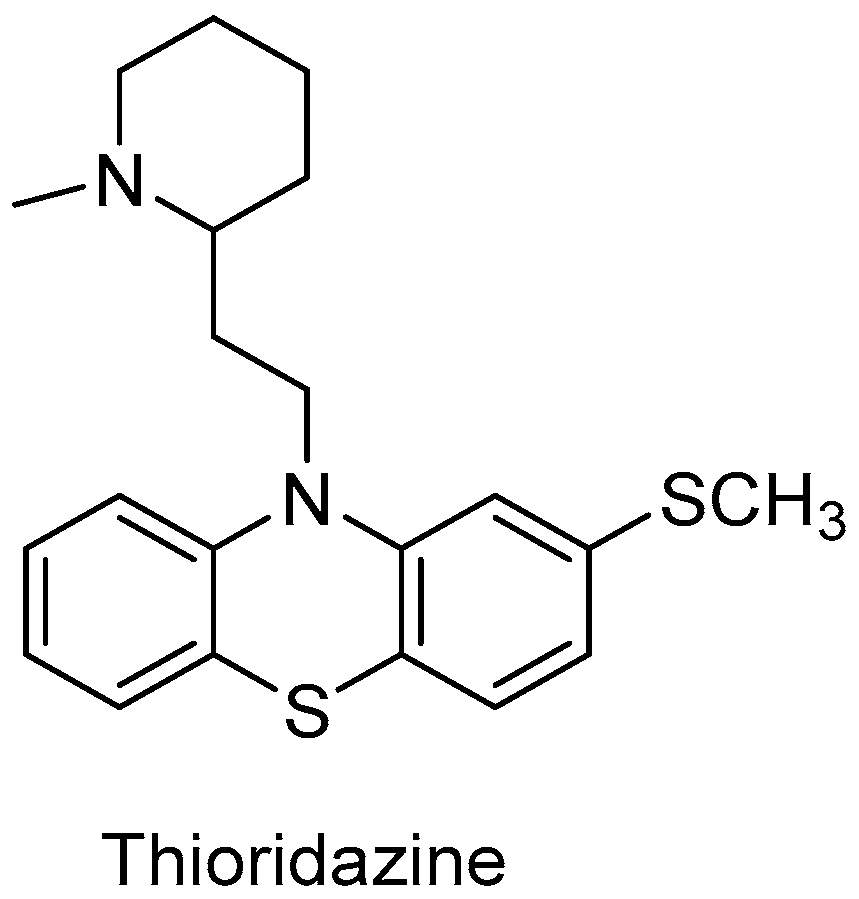
Catalysts | Free Full-Text | Biotechnological Methods of Sulfoxidation: Yesterday, Today, Tomorrow | HTML
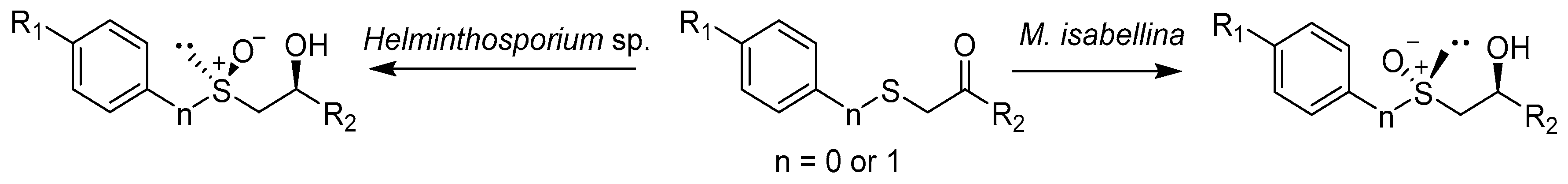
Catalysts | Free Full-Text | Biotechnological Methods of Sulfoxidation: Yesterday, Today, Tomorrow | HTML

Bond-Forming and -Breaking Reactions at Sulfur(IV): Sulfoxides, Sulfonium Salts, Sulfur Ylides, and Sulfinate Salts. - Abstract - Europe PMC

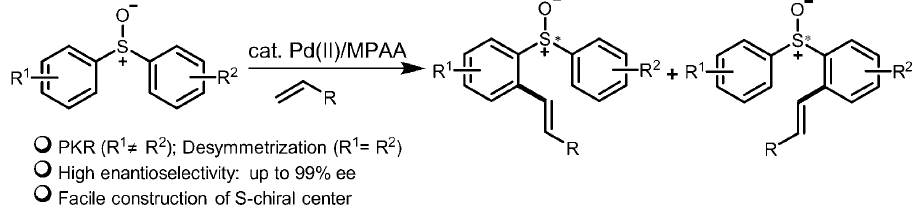
PPT - Preparation of Allyl Sulfoxides by Palladium-Catalyzed Allylic Alkylation of Sulfenate Anions PowerPoint Presentation - ID:3998393

Catalysts | Free Full-Text | Biotechnological Methods of Sulfoxidation: Yesterday, Today, Tomorrow | HTML
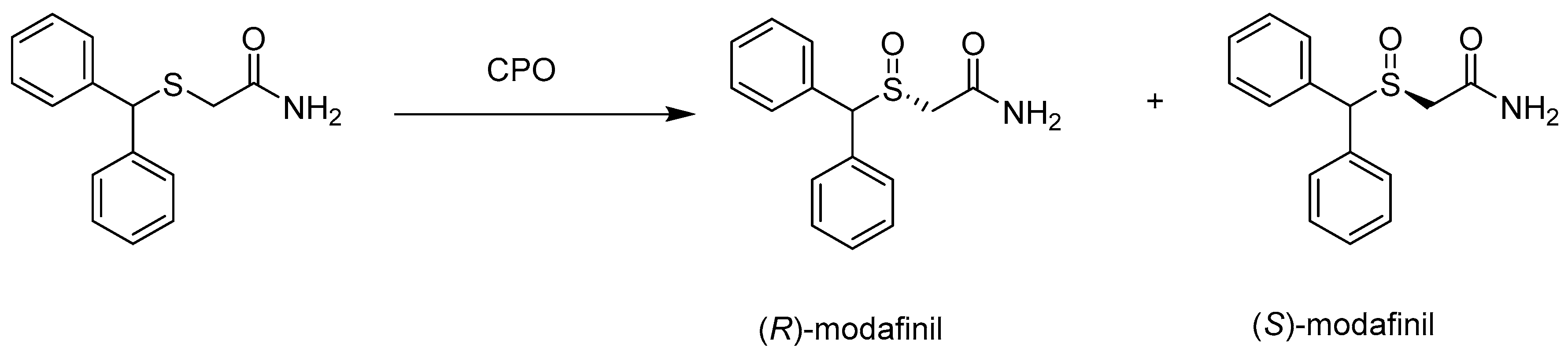
Catalysts | Free Full-Text | Biotechnological Methods of Sulfoxidation: Yesterday, Today, Tomorrow | HTML

Bis-phosphine monoxide platinum(II) and palladium(II) cationic complexes as Lewis acid catalysts in Diels–Alder and sulfoxidation reactions - ScienceDirect
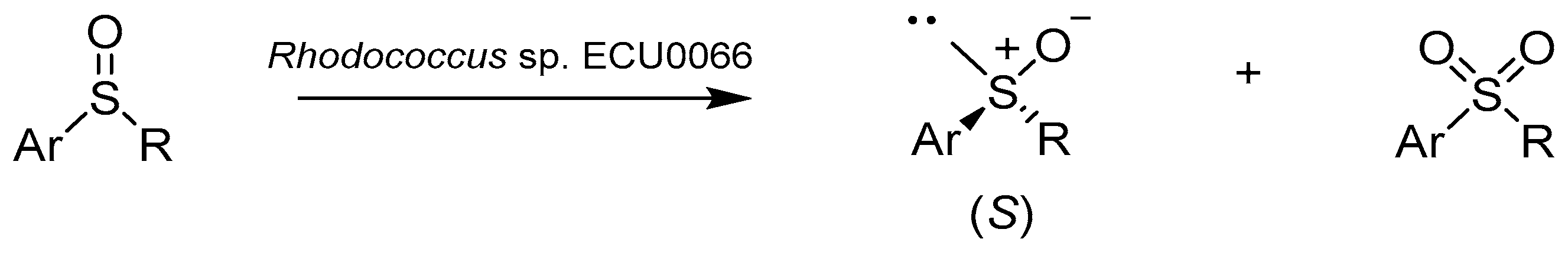
Catalysts | Free Full-Text | Biotechnological Methods of Sulfoxidation: Yesterday, Today, Tomorrow | HTML
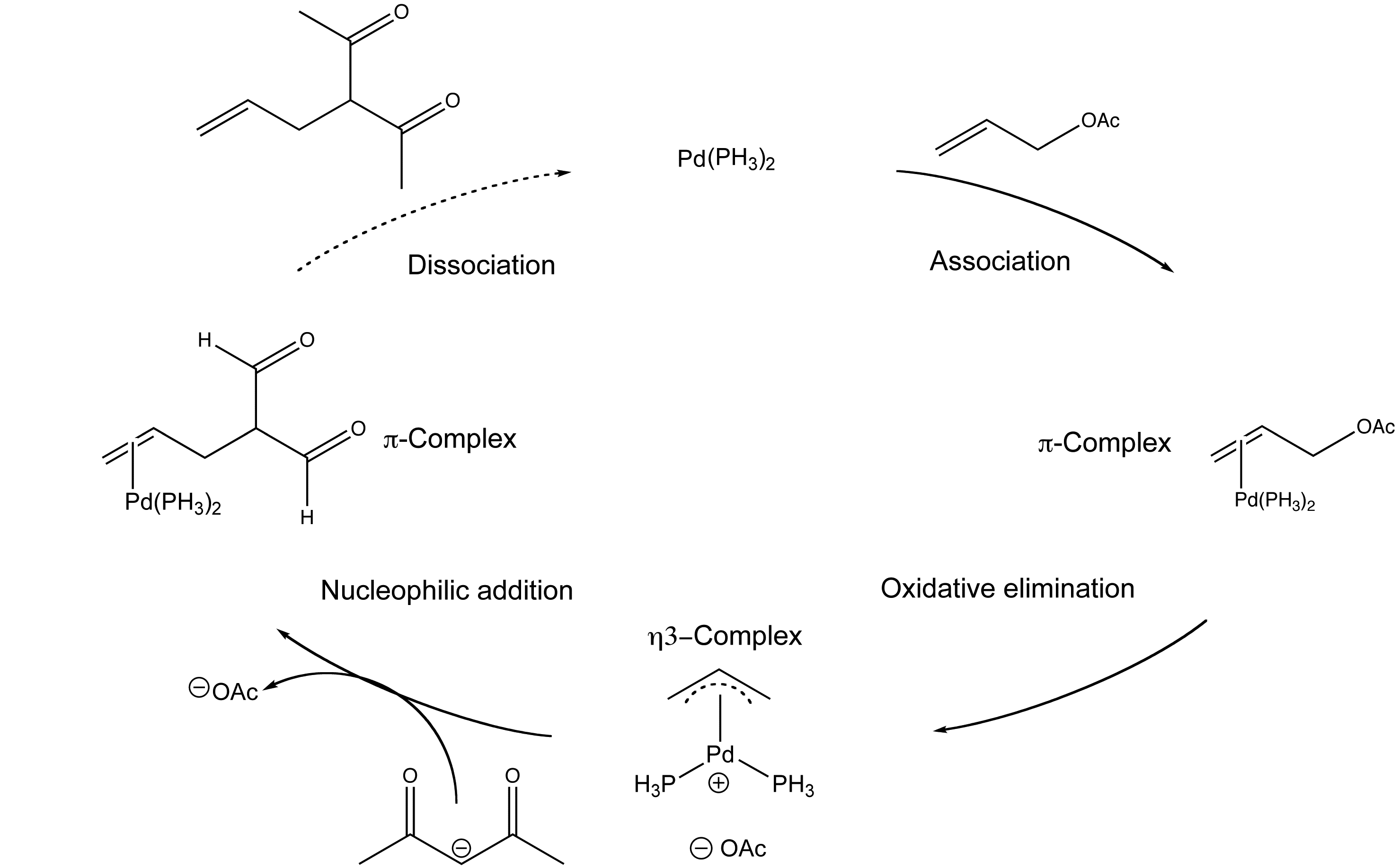
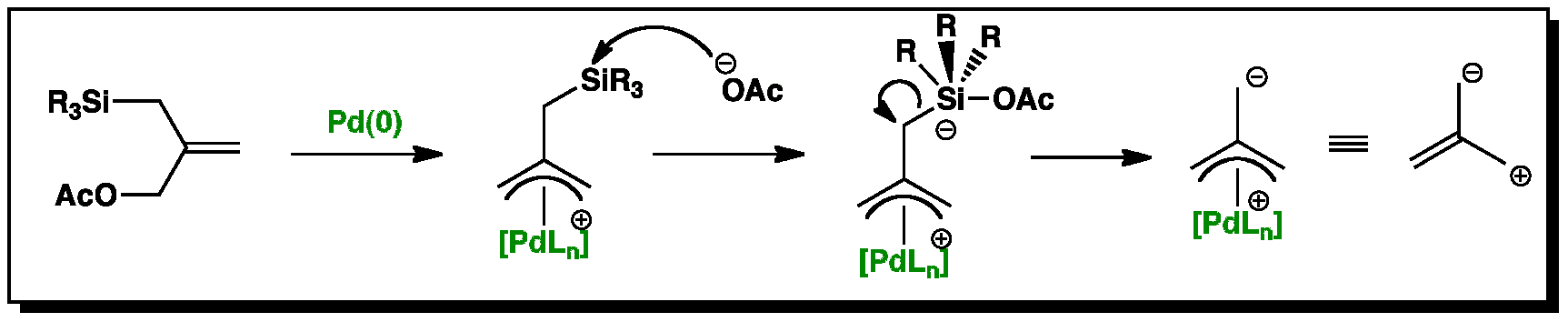
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds